Vacuum & Electrical Pulse Method for Plant Seed Electroporation
| Direct Gene Transfer into Mature Seeds via Electroporation |
| For the following Seeds:
– Rice – Rapeseed – Soyabean – Sorghum – Arabidopsis – Guinea Grass – Impatiens Balsamina – Green Chive – Plant – Wheat – Barley – Silk Worm, etc Photos of Seed Transformations with the NEPA21 system
|
Vacuum & Electrical Pulse
|
Advantages 1. Starting materials are mature seeds. 2. Transformed plants are obtained in a relatively short time. 3. Plant tissue culture is not necessary. 4. Possibly applicable to animal cells and microorganisms.
|
Basic Method
|
||
 |
 |
||
|
Vacuum Chamber & NEPA21/CUY21EDIT |
Chamber: CUY520P5 or P15 |
Electroporation on ice |
|
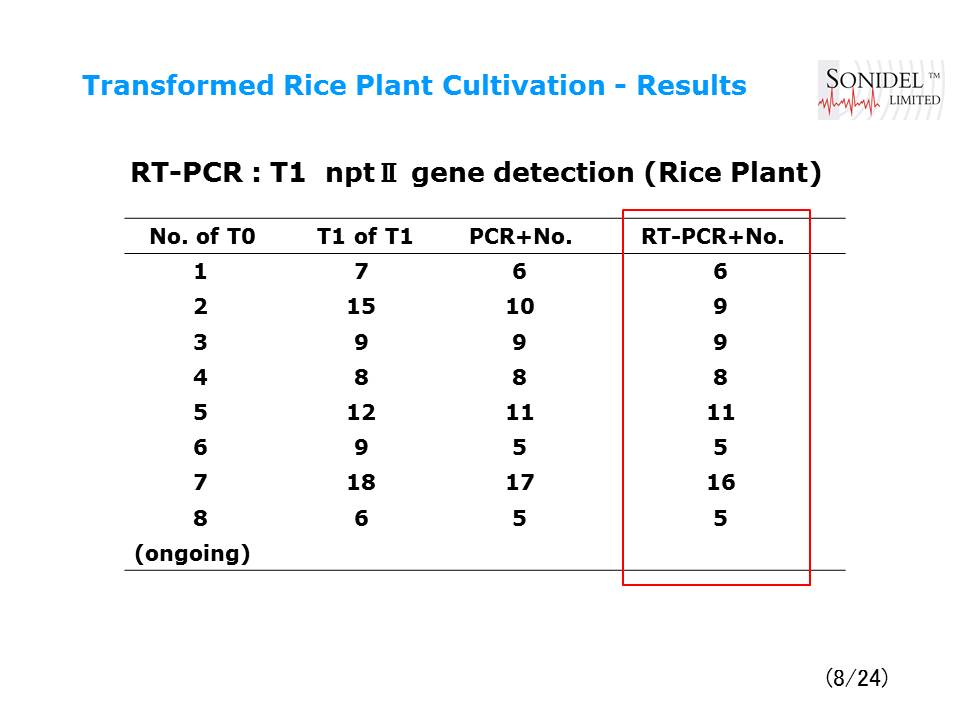
| Fertile transgenic plants were regenerated and self-fertilized seeds were obtained in rice and wheat. Transgene integration was confirmed by Southern hybridization. Transmission of the transgene into the next generation (T1) was indicated by PCR analysis. Transient gene expression was observed in several plant species and silk worm eggs. |
|
N: Negative control, –: Buffer only, V: Vacuum, E: Electroporation ………GUS gene expression reference (Wheat)The level of GUS gene expression by fluorescene assay (Wheat)
From left to right:
a) vacuum treatment and electroporation,
b) only vacuum treatment,
c) control GUS gene expression is visibly
observed in blue area
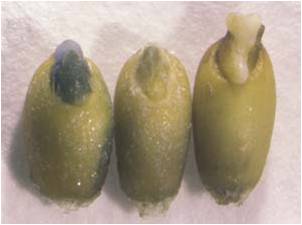
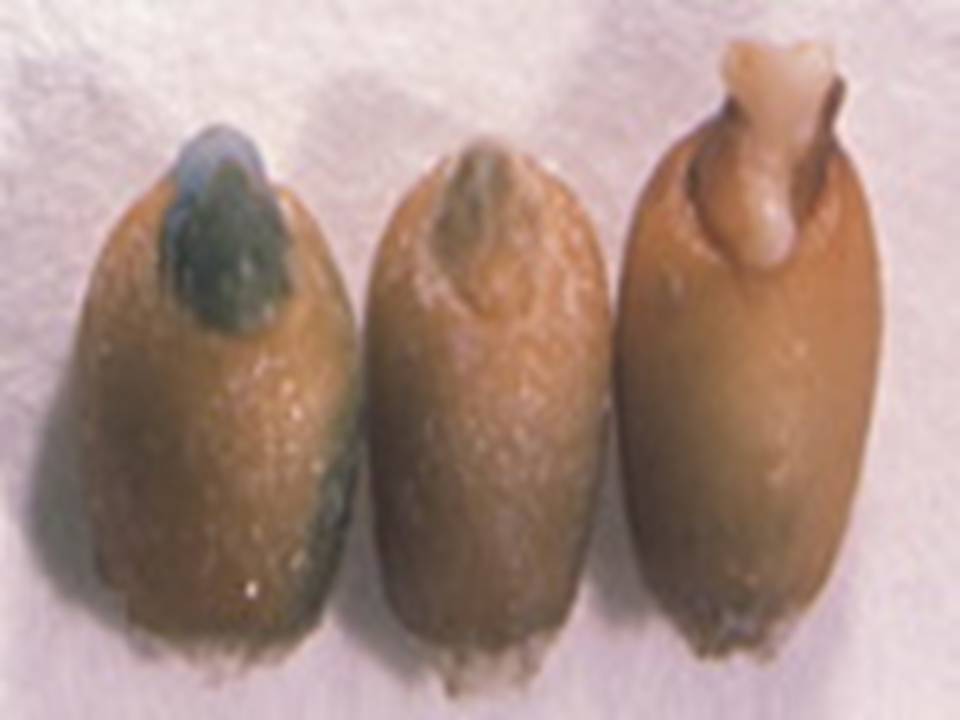
Not Transformed npt II gene introduced Next Generation (T1) Individual (Rice Plant)
Geneticine Selection of Rice Seeds and/or Seedings
Electroporation Apparatus and the Chamber |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
Gene transfer into Silk Worm Eggs
|
||||||||||||||||
| Plant Genetic Engineering Unit, National Institute of Agrobiological Sciences; Takashi Hagio * Improvement research is in progress in Dr. Hagio’s laboratory. | ||||||||||||||||
|
SUMMARY This system can directly deliver foreign genes into mature plant seeds using electroporation. Mature seeds of rice and wheat were soaked in water overnight. Then the seeds were incubated in an electroporation buffer containing plasmid DNAs for several hours under the condition of reduced air pressure. DNAs are considered to be transported into cells across cell walls with the help of depressurization treatment. Electroporation was carried out with a NEPA21/CUY21 electroporation device in a 10mm wide cuvette containing 1.0 ml of electroporation buffer. After the electroporation the seeds were incubated in water for two days. To determine the optimal gene transfer conditions, GUS (β-glucuronidase) gene expression assay were carried out. To obtain stably transformed plants, the seeds were transferred to a selection medium. After the plant regeneration putative transgenic chimeras and sexual offspring were analyzed. In both rice and wheat, fertile transgenic plants were regenerated and self-fertilized seeds were obtained. Transgene presence was confirmed by Southern hybridization. Transmission of the transgene into the next generation (T1) was indicated by PCR analysis. This method is simpler and more rapid than conventional techniques, and it can be applied to a wide range of commercial rice and wheat varieties. The transformation procedure presented here does not require the establishment of genotype-dependent tissue culture system. Transient gene expression in soybean seeds, tomato and Brassica were also observed. |
DOWNLOADS:
– Article: Direct Gene Transfer into Plant Mature Seeds via Electroporation After Vacuum Treatment
– Brochure: Direct Gene Transfer into Mature Seeds Via Electroporation
– Powerpoint: Gene Transfer System for Plant Seed, Egg and Microbe
CUY21 system for Gene Transfer System for Plant Seed, Egg & Mircobe
- Current Agrobacterium & Microinjection Methods – Disadvantages
- Vacuum & Electroporation Method – Advantages
- Vacuum & Electroporation Method – Basic Methodology Explained
- Vacuum & Electroporation Method – Apparatus Required
- Vacuum & Electroporation Methodology – Effectiveness
- Transformed Rice Plant Cultivation
- Transformed Rice Plant Cultivation – Results
- Gene Transfer to Silk Worm Eggs
- Gene Transfer to Arabidopsis Seed
- Gene Transfer to Yest
- New Vacuum & Electroporation Method – Improvement
If you click on e.g., ‘Transformed Rice Plant Cultivation – Results’ above, the following page will display:
 |
NEPA21 for Plant Seed Electroporation
Please note that the results illustrated above are for the efficiency of our old CUY21 system for wheat and rice.
As the NEPA21 was only recently launched we do not yet have comparable data for the NEPA21 system.
However, we have NEPA21 results data for sorghum seeds.
The CUY520P15 electrode was used to generated these results and is the recommended electrode.
For your further information:
– The NEPA21 system is the latest update to our old CUY21 system.
– The NEPA21 system can be set up to operate as a CUY21 system (so the illustrated CUY21 results can also be achieved with the NEPA21 system)
Related Information:
Plant Protoplast Fusion by Electro-Cell-Fusion and by Electroporation
Plant Seed Electroporation (For Seeds of Rice Plant, Wheat and Barley, Silk Worm, Yeast, etc)
Algae Transformation without cell-wall removal
ECFG21 device for Hybridoma Production/Nuclear Transfer