NEPA21_Retina_EP
NEPA21 Retina/Eye EP
In Vivo and Ex Vivo
For Application and Electroporation execution information on both Ex Vivo and In Vivo RETINA electroporation, please note the following link to our website:
Chapter 19 – Analysis of Gene Function in the Retina
Also of note are:
- Gene Transfer of Prolyl Hydroxylase Domain 2 Inhibits Hypoxiainducible Angiogenesis in a Model of Choroidal Neovascularization
Anna Takei, Malena Ekström*, Parviz Mammadzada*, Monica Aronsson, Ma Yu, Anders Kvanta & Helder André, 10 February 2017
The animal data is was generated with our NEPA21 system.
Koriyama Y1, Takagi Y, Chiba K, Yamazaki M, Sugitani K, Arai K, Suzuki H, Kato, August 2013
NEPA21 Retina/Eye EP
In Vivo and Ex Vivo
For Application and Electroporation execution information on both Ex Vivo and In Vivo RETINA electroporation, please note the following link to our website:
Chapter 19 – Analysis of Gene Function in the Retina
Also of note:
Gene Transfer of Prolyl Hydroxylase Domain 2 Inhibits Hypoxiainducible Angiogenesis in a Model of Choroidal Neovascularization
Anna Takei, Malena Ekström*, Parviz Mammadzada*, Monica Aronsson, Ma Yu, Anders Kvanta & Helder André, 10 February 2017
The animal data was generated with our NEPA21 system.
Requirement of Retinoic Acid Receptor b for Genipin Derivative-Induced Optic Nerve Regeneration in Adult Rat Retina
Koriyama Y1, Takagi Y, Chiba K, Yamazaki M, Sugitani K, Arai K, Suzuki H, Kato, August 2013
Retina – Ex Vivo EP
We recommend three possible electrode configurations for Retina Ex Vivo EP.
The retinas are dissected at embryonic stages (starting Embryonic day 12) and early postnatal for electroporation.
1) CUY700P5E and CUY700P5L
2) CUY701P7E and CUY700P5L (in some cases, a larger electrode at the bottom (positive electrode) works better).
3) CUY520P5 (See the protocol above: Chapter 19 – Analysis of Gene Function in the Retina).
Generally, we express a preference for the CUY520P5 electrode configuration because:
– it is the electrode configuration specified in detail in the publication: Chapter 19 – Analysis of Gene Function in the Retina,
.
Ex Vivo Retina Photo
(CUY520P5 Configuration)

Ex Vivo Retina Photo
(CUY701P2e / CUYP2L Configuration)
 |
CUY701P2E / CUY701P2L |
|
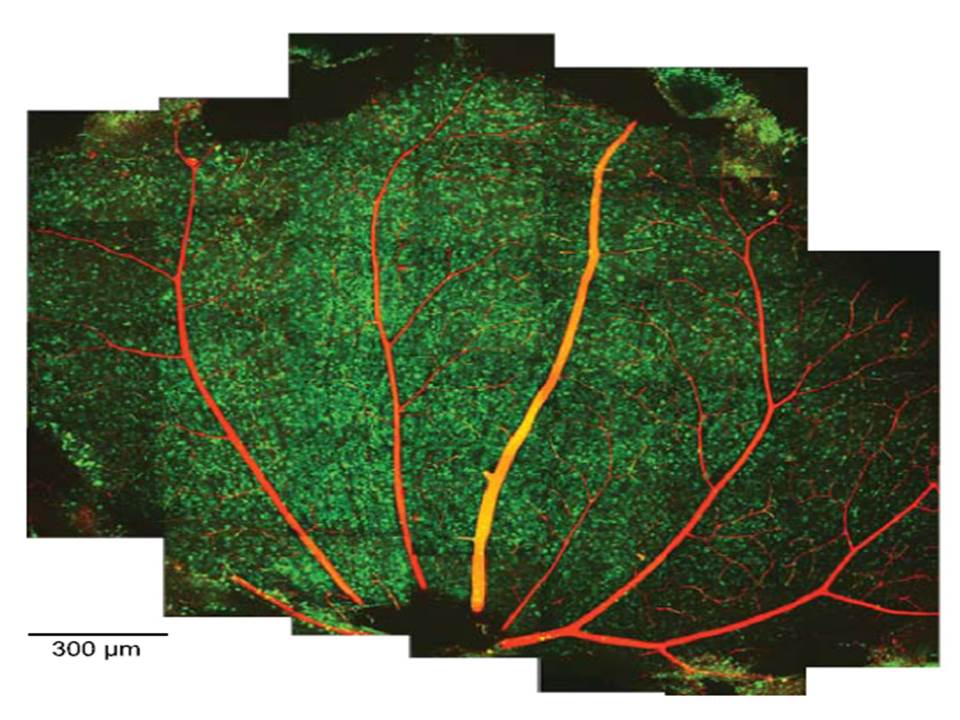 |
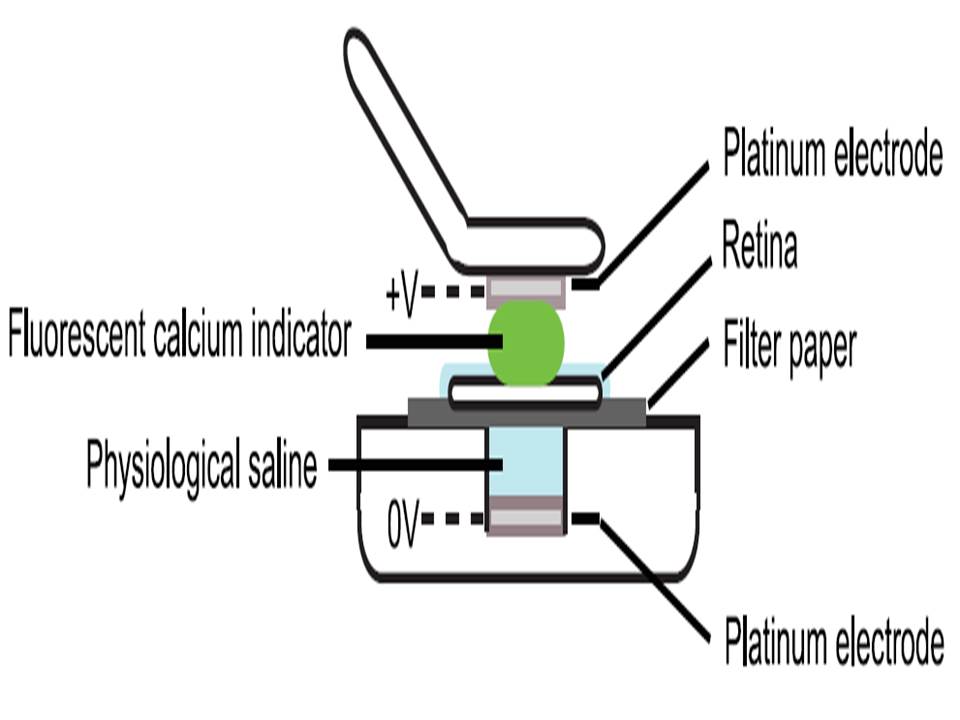
Montage of 2-photon (2P) micrographs of whole-mounted mouse retina Green: Oregon Green 488 BAPTA-1 [OGB-1; focal plane in ganglion cell layer (GCL), only green channel shown] Red: sulforhodamine 101 (SR101) |
|
| Briggman KL, et al.,Bulk electroporation and population calcium imaging in the adult mammalian retina., J Neurophysiol. 2011 May;105(5):2601-9 | ||
Retina – In Vivo / Post Natal EP
 |

CUY675P5 |
.. |  |

CUY650P7 |
 |
 |
|||
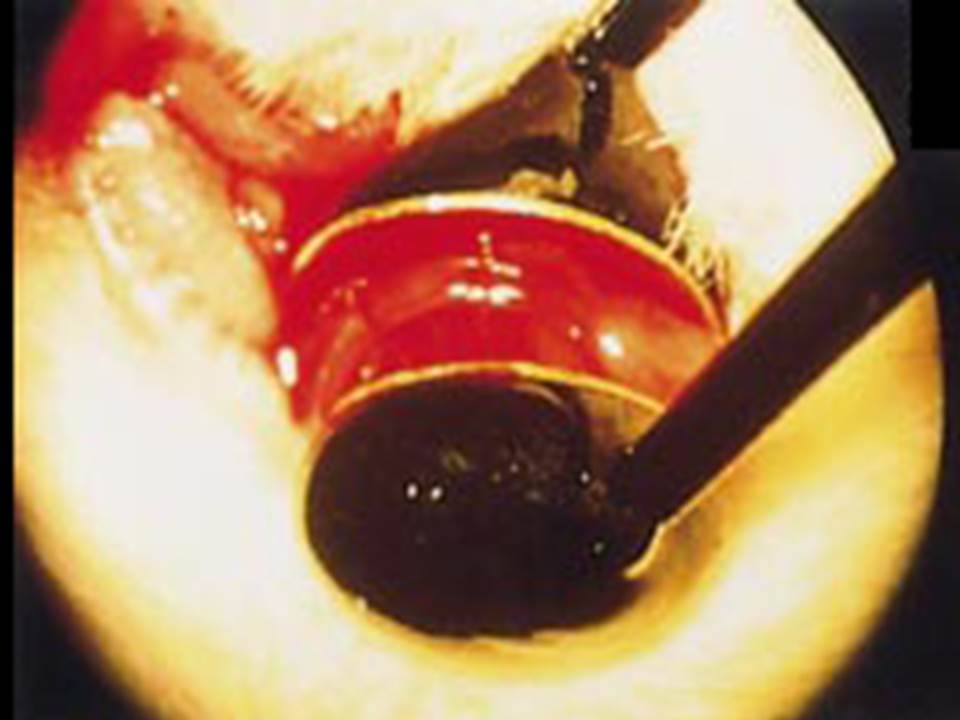
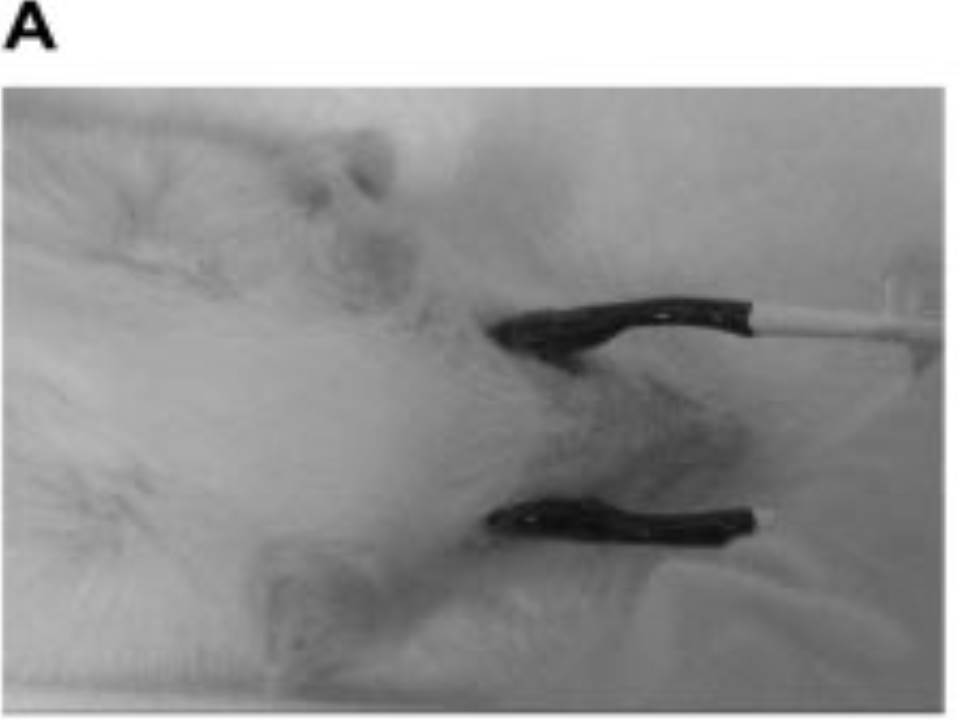
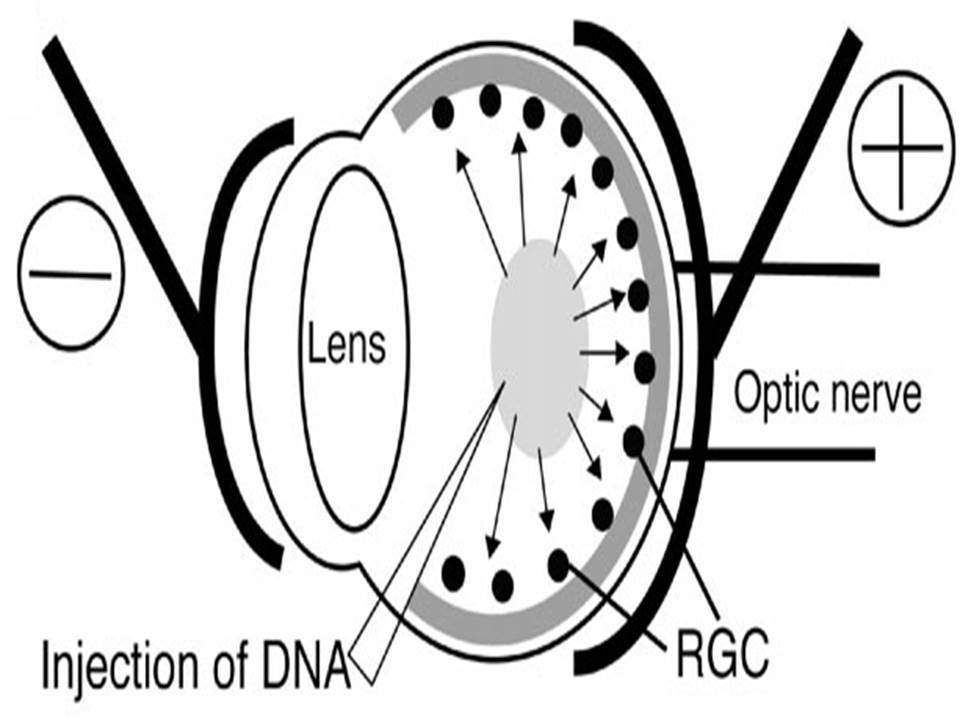
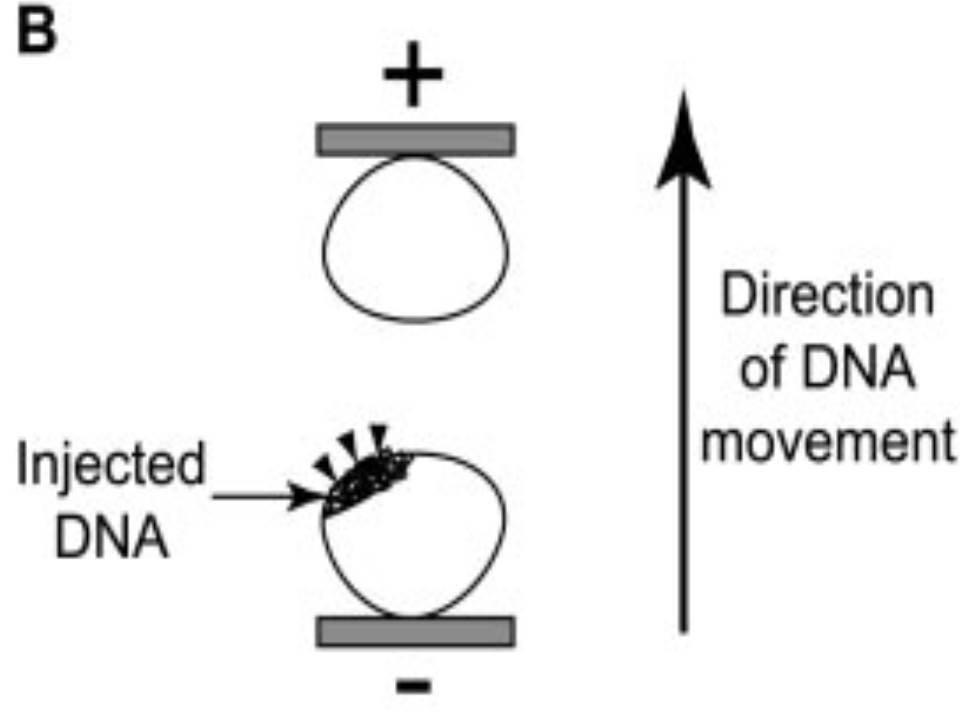
| The left eye and the optic nerve were exposed.
A small hole was made with a 31-gauge needle 0.5 to 1.0 mm The eye was then immediately grasped between tweezerz-type electrodes, with the cathode attached to the corneal electrode and the anode with the slit attached to the scleral electrode. |
Arrangement of electrodes for in vivo electroporation for RPE transfection.
(A) Tweezer-type electrodes were placed on the corneal surface of either eye of a 1-month-old Sprague-Dawley rat. (B) The current was applied with the positive electrode contralateral to the injected eye. After prior injection of plasmid DNA into the subretinal space of the right eye, this arrangement electrophoresed the negatively-charged DNA toward the RPE layer (arrowheads). |
|||
We recommend both the CUY675P5 and CUY650P7 electrodes for In vivo/postal natal Retina EP.
However, most clients opt for the CUY650P7 configuration because it can also be used for (whole) Cultured Embryonic (and Large Tissue) EP.
– Cultured Mouse Embryos and Large Tissue EP
In Vivo / Ex Vivo / Whole Embryo Culture EP Publications and Electrode Recommendations
|
Target |
Publication |
Electrode |
Protocol . |
| Mice/Rats Cultured Embryos |
Koike S, Keino-Masu K, Ohto T, Sugiyama F, Takahashi S, Masu M. Autotaxin/lysophospholipase D-mediated lysophosphatidic acid signaling is required to form distinctive large lysosomes in the visceral endoderm cells of the mouse yolk sac. J Biol Chem. 2009 Nov 27;284(48):33561-70. Epub 2009 Oct 5. |
CUY650P10 | Ask Us |
| Okada T, Okumura Y, Motoyama J, Ogawa M. FGF8 signaling patterns the telencephalic midline by regulating putative key factors of midline development. Dev Biol. 2008 Aug 1;320(1):92-101. Epub 2008 May 8. |
CUY666P0.3 | Ask Us | |
| Shibuya et al. Isolation and Characterization of Vasohibin-2 as a Homologue of VEGF-Inducible Endothelium-Derived Angiogenesis Inhibitor Vasohibin Arteriosclerosis, Thrombosis, and Vascular Biology, Volume 26, Issue 5, Pages1051-1057, May 2006 |
CUY520P series | Ask Us | |
| Tadashi Nomura and Noriko Osumi Misrouting of mitral cell progenitors in the Pax6/small eye rat telencephalon Development, Volume 131, Issue 4, Pages 787-796, February 2004 |
CUY650P7 | Ask Us | |
| Masanori Takahashi and Noriko Osumi Pax6 regulates specification of ventral neurone subtypes in the hindbrain by establishing progenitor domains Development, Volume 129, Issue 6, Pages 1327-1338, March 2002 |
CUY520P20 | Ask Us | |
| Takahashi et al. Manipulating gene expressions by electroporation in the developing brain of mammalian embryos Differentiation, Volume 70, Issue 4-5, Pages 155-162, June 2002 |
CUY520P20 CUY650P7 |
Ask Us | |
| Noriko Osumi and Takayoshi Inoue Gene Transfer into Cultured Mammalian Embryos by Electroporation Methods, Volume 24, Issue 1, Pages 35-42, May 2001 |
CUY520P series CUY650P series |
Ask Us | |
| Mice/Rats in vivo/ex vivo Retina |
O Ros, Xavier Nicol et al
SponGee: a genetic tool for subcellular and cell-specific CGMP manipulation Cell Rep. 2019 Jun 25;27(13):4003-4012.e6. doi: 10.1016/j.celrep.2019.05.102. |
Ask Us | |
| Takei A, Ekström M, Mammadzada P, Aronsson M, Yu M, Kvanta A, André H | CUY650P5 | Ask Us | |
| Koriyama Y, Takagi Y, Chiba K, Yamazaki M, Sugitani K, Arai K, Suzuki H, Kato S. Requirement of retinoic acid receptor β for genipin derivative-induced optic nerve regeneration in adult rat retina PLoS One. 2013 Aug 6;8(8):e71252. |
CUY650P | Ask Us | |
| Tracy L. Young and Constance L. Cepko A role for ligand-gated ion channels in rod photoreceptor development Neuron, Volume 41, Issue 6, Pages 867-879, 25 March 2004 |
Vitro: CUY532 Vivo: as described (Matsuda et al. 2004) |
Ask Us | |
| Takahiko Matsuda and Constance L. Cepko Electroporation and RNA interference in the rodent retina in vivo and in vitro PNAS, Volume 101, Number 1, Pages 16-22, 6 January 2004 |
Vivo: CUY650P7 Vitro: CUY520P5 |
Ask Us | |
| Dezawa et al. Gene transfer into retinal ganglion cells by in vivo electroporation: a new approach Micron Volume 33, Issue 1, Pages 1-6, 2002 |
CUY675P3 CUY675P5 |
Ask Us | |
Mice/Rats ex vivo Retina |
Benjamin Souferi, Mark M Emerson
Quantitative Analysis of the ThrbCRM1-centered Gene Regulatory Network Biol Open,8 (4) 2019 Apr 26 |
||
| Mark M Emerson, Constance L Cepko
Dev Biol, 360 (1), 241-55 2011 Dec 1 |
CUY700P3E
CUY700P3L |
||
| Briggman KL, Euler T. Bulk electroporation and population calcium imaging in the adult mammalian retina J Neurophysiol. 2011 May;105(5):2601-9. Epub 2011 Feb 23. |
CUY700P3E CUY700P3L |
Ask Us | |
| Ozawa Y et al. Roles of STAT3/SOCS3 pathway in regulating the visual function and ubiquitin-proteasome-dependent degradation of rhodopsin during retinal inflammation J Biol Chem. 2008 Sep 5;283(36):24561-70. Epub 2008 Jul 9. |
CUY700P series | Ask Us | |
| Mice/Rats in vivo Cornea |
Oshima et al. Targeted Gene Transfer to Corneal Stroma in vivo by Electric Pulses Experimental Eye Research, Volume 74, Issue 2, Pages 191-198, February 2002 |
CUY671P1 | Ask Us |
| Chick
Embryos in ovo |
Estie Schick, Sean D McCaffery, Erin E Keblish, Cassandra Thakurdin, Mark M Emerson
Sci Rep, 9 (1), 9358 2019 June 27 |
||
| Miruna G Ghinia Tegla, Diego F Buenaventura, Diana Y Kim, Cassandra Thakurdin, Kevin G Gonzalez, Mark M Emerson
bioRxiv. 2019 Februarys 2, https://dio.org/10.1101/538710 |
|||
| Jin et al.
Development, Volume 130, Issue 6, Pages 1037-1048, March 2003 |
CUY611 series
CUY614 & CUY615 |
||
| Chicken Embryos ex ovo | Miruna G Ghinia Tegla, Diego F Buenaventura, Diana Y Kim, Cassandra Thakurdin, Kevin G Gonzalez, Mark M Emerson
bioRxiv. 2019 Februarys 2, https://dio.org/10.1101/538710 |
|
| Chicken Retina
ex vivo |
Estie Schick, Sean D McCaffery, Erin E Keblish, Cassandra Thakurdin, Mark M Emerson
Sci Rep, 9 (1), 9358 2019 June 27 |
||
| Zebrafish
Retina In vivo / ex vivo |
Kustermann et al.,
Cell and Tissue Research, Volume 332, Number 2, Pages 195-209, May 2008 |
CUY550-10
CUY701P2E |
| Thummel et al.,
Inhibition of Müller glial cell division blocks regeneration of the light-damaged zebrafish retina Developmental Neurobiology, Volume 68, Issue 3, Pages 392-408, 15 February 2008 |
CUY650P3 |
| Cell Cultures (adherent electroporation)Retinal Ganglion Cells from human iPSCs |
Tanaka T, Yokoi T, Tamalu F, Watanabe S, Nishina S, Azuma N Generation of retinal ganglion cells with functional axons from human induced pluripotent stem cells. Sci Rep. 2015 Feb 10; 5: 8344. |
Other relevant links to our website
– Zebrafish Retina EP
– Protocol for In Vivo Mouse and Rat Retina EP
– KnowHow
In H. Nakamura (ed.), Electroporation and Sonoporation in Developmental Biology, 143
DOI: 10.1007/978-4-431-09427-2_14, © Springer 2009
‘Chapter 14: In Utero Electroporation: Assay System for Migration of Cerebral Cortical Neurons‘
Hidenori Tabata and Kazunori Nakajima
– Mouse/Rate Embryo – Eye
– Gene transfer into embryonic brains using In Utero EP Technique
– Cultured Mouse Embryos and Large Tissue EP
==============
==============
==============
Early Postnatal Mouse Eye/Retina and In Utero Embryo EP
Early Postnatal Mouse Eye/Retina EP
| For your information:
With respect to gene delivery into Mouse Embryo Eye/Retina (and Brain) one can use the tungsten needle type electrode. However, as the mouse embryo is very small it is often hard to inject DNA into the embryo’s eye. Some clients have reported using the CUY675P5 electrode It is structurally possible to use this electrode for both Embryo (in utero) and Mouse Eye electroporation. (Please note the table below).
As one can see, however, there is a cut-out on one side of the CUY675P type electrodes. So, for this application we are not convinced that it is the optimum electrode for the application. Rather, our recommendation is for the CUY650P7 electrode. Note the attached CUY650P7 Technical Drawing.
This is the electrode most of our clients choose. It has a fully rounded set of electrode paddles and therefore covers a larger area of the target. In addition, this electrode has a much wider application range and can be used, for example, for or (whole) Cultured Mouse Embryos and Large Tissue EP. Dr. Matsuda uses the CUY650P7 electrode in his articles contained in the ‘EP with Retina’ below). Takahiko Matsuda and Constance L. Cepko Electroporation and RNA interference in the rodent retina in vivo and in vitro PNAS, Volume 101, Number 1, Pages 16-22, 6 January 2004 |
||||||||||||||||
In Utero Embryo EP
| The standard electrodes for In Utero Embryo EP are CUY650P3 and CUY650P5, which have circle electrodes without a cut. |
Mouse Embryo – Cornea
| For gene delivery into Mouse Embryo – Cornea we recommend:
– CUY671P1 or Technical Drawing: CUY671P1 – CUY670 Technical Drawing: CUY670 Protocol Recommendation: – ‘Electroporation and RNA interference in the rodent retina in vivo and in vitro‘ – ‘Protocol for in vivo electroporation into mouse/rat retina’ – ‘Compensation by tumor suppressor genes during retinal development in mice and humans‘ For your further information, note the article titled: – ‘A role for ligand-gated ion channels in rod photoreceptor development’. In this article and ‘Electroporation and RNA interference in the rodent retina in vivo and in vitro’ above genes are transfected into mouse and rat retina. We believe that the method outlined in these articles can be applied to Gene Therapy |
Retina Cell EP – Results
|
|||||||||||||||||||||||||||
(See the cell images by clicking the cell names)
V: Viability, TE: Transfection Efficiency
A larger range of successfully electroporated cells is available at this link to our website: SONIDEL Cell Transfection Database

